| STANNOUS FLUOBORATE | |||
|
PRODUCT IDENTIFICATION |
|||
| CAS NO. | 13814-97-6 |
| |
| EINECS NO. |
237-487-6 | ||
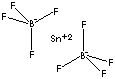
| FORMULA | Sn(BF4)2 | ||
| MOL WT. | 292.30 | ||
|
H.S. CODE |
2826.90 | ||
| TOXICITY | |||
| SYNONYMS | Tin Fluoroborate; Stannous fluoroborate; | ||
| Tin(II) tetrafluoroborate; Zinnbis(tetrafluoroborat) (Greman); Bis(tetrafluoroborato) de estaño (Spanish); Bis(tétrafluoroborate) d'étain (French); | |||
| SMILES | |||
|
CLASSIFICATION |
|
||
|
PHYSICAL AND CHEMICAL PROPERTIES (50% SOLUTION) |
|||
| PHYSICAL STATE |
Clear Solution | ||
| MELTING POINT |
| ||
| BOILING POINT | |||
| SPECIFIC GRAVITY | 1.67 | ||
| SOLUBILITY IN WATER | Readily soluble | ||
| pH | 4.5 - 6.5 | ||
| VAPOR DENSITY | |||
| AUTOIGNITION |
| ||
| NFPA RATINGS |
| ||
| REFRACTIVE INDEX |
| ||
| FLASH POINT | Not considered to be a fire hazard | ||
| STABILITY | Stable under ordinary conditions. | ||
|
GENERAL DESCRIPTION & APPLICATIONS |
|||
|
Application:Fluoroboric acid is used in plating circuits; metal finishing; electropolishing of aluminium and its alloy; component of galvanic baths; organic synthesis as catalyst for alkylations and polymerisation; stabilisation of diazo salts; manufacturing of inorganic fluoroborate salts. Inorganic fluoroborate salts are used as components of fluxing and plating, as catalysts, in flame-retardant manufacture, in metal treatment; grain refining agents; as active fillers in resin bonded abrasives; in electrolytic generation of boron, preparation of glazing frits. |
|||
| SALES SPECIFICATION | |||
|
APPEARANCE |
Clear Solution | ||
| ASSAY Sn(BF4)2 | 50-52 | ||
| FREE BORIC ACID | 2.0% max | ||
| FREE FLUOBORIC ACID | 3.0% max | ||
| STANNIC TIN (Sn) | 1.0% max | ||
| LEAD (Pb) | 0.0% max | ||
| CHLORIDE (Cl) | 0.005% max | ||
| SULFATE (SO4) | 0.03 max | ||
| IRON (Fe) |
0.01% max | ||
| COPPER (Cu) |
0.005% max | ||
| ZINC (Zn) | 0.001% max | ||
| NICKEL (Ni) | 0.001% max | ||
| COLOR (APHA) | 200 max | ||
| TRANSPORTATION | |||
| PACKING | 300kgs in Drum | ||
| HAZARD CLASS | 8 (Packing Group: III) | ||
| UN NO. | 3264 | ||
| OTHER INFORMATION | |||
| Hazard Symbols: C, Risk Phrases: 34, Safety Phrases: 26-36/37/39-45 | |||
|
GENERAL DESCRIPTION OF TIN COMPOUNDS |
|||
|
Tin compounds are classified into two main groups; inorganic-tin and organo-tin compounds. The organo-tin compounds are defined as compounds in which at least one tin-to-carbon bond exist. But the inorganic-tin compounds do not contain carbon as the principal element. Inorganic-tin compounds are relatively simple in their molecular structure and, like tin itself, are not considered to be toxic. Tin atoms can replace carbon atoms in chemical compounds, and a great variety of organo-tin compounds are known. INORGANIC TIN COMPOUNDS The largest use for inorganic tin compounds is in electrolytes for plating tin and tin alloys. The more important plating chemicals are chlorides, sulfates, and fluoroborates in acidic electrolytes and stannates in alkaline solutions. Inorganic-tin compounds are divided into two series: stannous, or tin(II), compounds and stannic, or tin(IV), compounds.Chemically, tin exhibits valencies of 2 and 4. It resists attack by water but is dissolved by strong acids and alkalis. One of common compounds of tin(II) are stannous chloride (SnCl2) used in tin galvanizing, as a reducing agent in the manufacture of polymers and as a mordant in dyeing.; stannous oxide (SnO) employed in making tin salts for chemical reagents and for plating; and stannous fluoride (SnF2) is the additive in fluoride tooth-pastes. Inorganic tin chemicals are used as catalysts in a number of industrial processes. stannous octoate is the catalyst that produces the foaming action that turns the liquid plastic into a foamlike solid structure in the manufacture of polyurethane foam. Tin(IV) compounds of significance include stannic chloride (SnCl4) is widely used as a stabilizer for perfumes and as a starting material for other tin salts; and stannic oxide(SnO2) is a useful catalyst in certain industrial processes and a polishing powder for steel. Tin sulfide is used as a bronzing agent for wood colouring ORGANOTIN COMPOUNDS The greatest use of di-organotin compounds is stabilizers in the
manufacture of polyvinyl chloride, or PVC. The particular importance of these
di-organotins lies in their outstanding ability to preserve the clarity and
transparency of PVC, not only when being processed but also in subsequent
service. Organotin-stabilized PVC is used in water pipes and in food packaging
applications as tin compounds used in these applications are known as nontoxic.
In contrast to the nontoxic compounds employed as stabilizers, some
tri-organotin compounds (e.g., tributyl- and triphenyltins) are powerful
biocides and have found use in a number of relevant applications, such as
fungicide, algicide, molluscicide in underwater and anti-fouling paints
extensively, preservatives for wood, as slimicide on masonry, as biocide
disinfectant for textile and leather processing, cooling system, pulp and paper
mill and brewery. The tributyltin family or fentine (triphenyltin) chemicals
include;
Tributyltin compounds are usually clear to yellowish liquids with an unpleasant odor. Triphenyltincompounds are white solids with low vapour pressures. Tri-organotin compounds are derivatives of tetravalent tin. They are lipophilic and have low water solubility. Physical and chemical properties of tri-organotin compounds vary depending upon the anion linked to tin. Tributyltin derivatives have toxic properties to gram positive bacteria are used as disinfectants on surfaces such as hospital floors and sports arenas, combined with gram negative bactericides. Tin chemicals also used as flame retardants to treat fabrics and plastics. Tributyltin methacrylate is used as a stabiliser for PVC. Other industrial applications of organotin compounds include as rodent repellents, antioxidants, curing agents and corrosion inhibitors. |
|||
| PRICE INFORMATION | |||